- Introduction: From Inspection to Prediction
In the framework of ISOSAF, Quality Control (QC) is the operational heartbeat of the “Assessment” pillar. While Quality Assurance (QA) focuses on the process, Quality Control focuses on the product. However, modern industrial science has moved beyond simple “pass/fail” inspections. The integration of Statistical Process Control (SPC) allows organizations to shift from reactive detection to proactive prevention. This article explores the mathematical and legal rigor required to maintain product integrity in high-stakes industries like pharmaceuticals and precision engineering.
- The Scientific Foundation of Quality Control
Quality Control is rooted in the scientific method: observation, hypothesis, testing, and verification. In a laboratory or manufacturing setting, QC involves a series of analytical procedures designed to ensure that a finished product meets its pre-defined specifications.
- Analytical Chemistry: Utilizing instruments like HPLC, GC-MS, and ICP-OES to verify chemical purity.
- Microbiology: Testing for pathogens or bioburdens to ensure safety.
- Physical Testing: Measuring dimensions, hardness, or stress tolerance with micron-level precision.
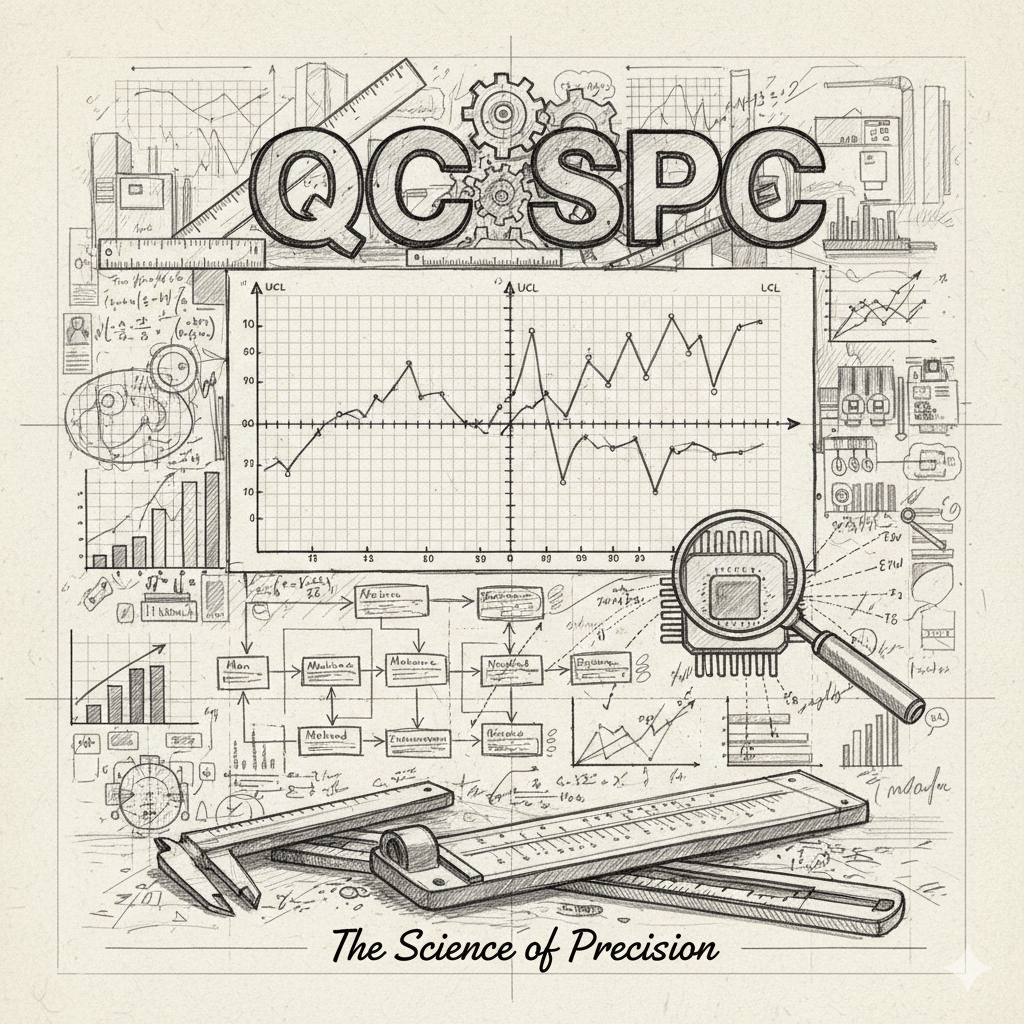
- Statistical Process Control (SPC): The Mathematical Compass
SPC is the application of statistical methods to monitor and control a process. It was pioneered by Walter Shewhart and further developed by W. Edwards Deming. The core philosophy is that every process contains variation.
- Common Cause Variation: Inherent to the process (e.g., slight vibrations in a machine).
- Special Cause Variation: External factors that signal a problem (e.g., a broken tool or a change in raw material quality).
- The Toolset of Precision: Control Charts and Capability Indices
To manage variation, engineers use several critical tools:
- Control Charts (Shewhart Charts): These plot data points over time against Upper Control Limits (UCL) and Lower Control Limits (LCL). If data points drift toward a limit or exhibit a non-random pattern, the process is “out of control” even if the products are still within legal specifications.
- Process Capability Index (Cpk): This is a scientific formula that measures how close a process is running to its specification limits, relative to the natural variability of the process. A higher Cpk (e.g., 1.33 or 1.67) indicates a highly stable and capable process.
- Six Sigma Methodology: A data-driven approach aimed at reducing defects to 3.4 per million opportunities.
- Legal Implications of QC Failure
From a legal perspective, QC is the primary defense against product liability. Under the “Strict Liability” doctrine in many international jurisdictions, a manufacturer is liable for damages caused by a defective product regardless of intent.
- Due Diligence: Robust SPC records provide a legal “paper trail” proving that the manufacturer maintained the “Standard of Care.”
- Admissibility: For results to be legally binding, the QC laboratory must be accredited under ISO/IEC 17025, ensuring that the equipment is calibrated and the methods are validated.
- Sampling Theory and Risk Management
It is often scientifically and economically impossible to test 100% of products (e.g., destructive testing). Therefore, QC relies on Statistical Sampling.
- AQL (Acceptable Quality Level): The maximum percentage of defects that is considered acceptable for a given batch.
- Legal Risks of Sampling: Choosing an incorrect sampling plan (e.g., n=3 for a batch of 10,000) can lead to “Type II Error”—accepting a bad batch—which can result in massive recalls and regulatory fines.
- Integrating QC into the ISOSAF “Frontier Development”
At ISOSAF, we lead the transition toward Quality 4.0. This involves using the “Observation” pillar to feed real-time sensor data into AI algorithms.
- Real-time Release Testing (RTRT): Instead of waiting days for lab results, sensors monitor critical process parameters (CPPs) to ensure quality during production.
- Digital Twins: Creating a virtual model of the production line to simulate QC outcomes before a single physical unit is produced.
- The Human Element: Training and Ethics
Despite the high-tech tools, QC remains a human endeavor. The “Frontier Development” of ISOSAF emphasizes the training of auditors and technicians. Ethically, a QC technician must have the legal and organizational authority to “stop the line”—a principle known in Japanese manufacturing as Jidoka.
- Conclusion: Precision as a Competitive Advantage
Quality Control and SPC are the guardians of brand reputation and consumer safety. For enterprises in Vietnam, China, and Singapore, mastering these scientific tools is the only way to compete in the high-value global export market. As our Technical Director, Dr. Klaus Schmidt, often emphasizes: “We do not guess; we measure. Precision is the language of trust.”
- References and Reliable Sources
- Montgomery, D. C. (2019). Introduction to Statistical Quality Control. 8th Edition, Wiley.
- ISO (2017). ISO/IEC 17025:2017 General requirements for the competence of testing and calibration laboratories.
- Wheeler, D. J., & Chambers, D. S. (1992). Understanding Statistical Process Control. SPC Press.
- Juran, J. M., & De Feo, J. A. (2010). Juran’s Quality Handbook: The Complete Guide to Performance Excellence. McGraw-Hill.
- International Council for Harmonisation (ICH). ICH Q10: Pharmaceutical Quality System.
- American Society for Quality (ASQ). Statistical Process Control (SPC) Reference Manual.
- ISOSAF Technical Bulletin. Implementing Cpk Analysis in Emerging Manufacturing Markets.